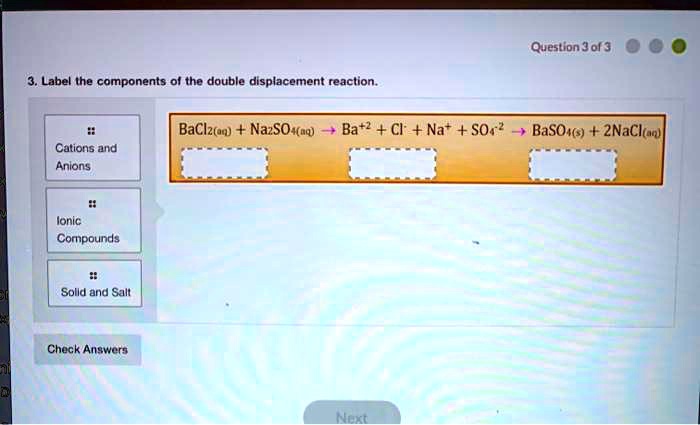
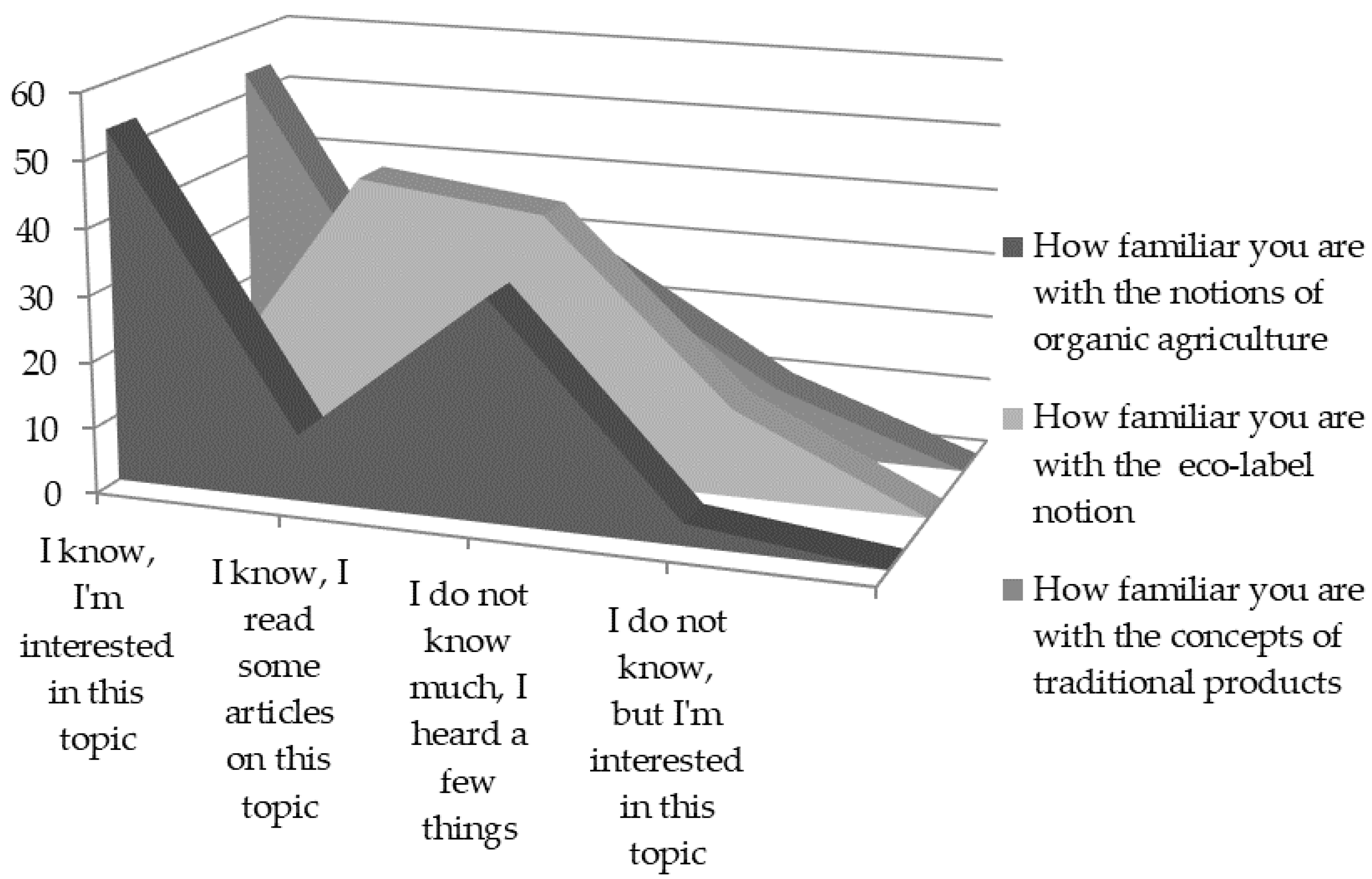
42 label the components of the double displacement reaction.
CH150: Chapter 5 - Chemical Reactions - Chemistry Double replacement reactions can be of two major types: (1) precipitation reactions wherein, at least one of the products forms a solid precipitate during the reaction, and (2) acid-base neutralization reactions, wherein an acid is neutralized by a base to form a salt and water. The solubility rules are used to determine if products in a double ... Types of Chemical Reactions - Detailed Explanation With Example ... - BYJUS Double Displacement Reaction A chemical reaction in which ions get exchanged between two reactants which form a new compound is called a double displacement reaction. It takes the form of XY + ZA → XZ + YA It is also called a metathesis reaction Example of double displacement reaction: BaCl 2 + Na 2 SO 4 → BaSO 4 + 2NaCl 5. Precipitation Reaction
Double replacement reactions - Khan Academy Double replacement reactions have two ionic compounds that are exchanging anions or cations. Precipitation reactions and neutralization reactions are two common types of double replacement reactions. Precipitation reactions produce an insoluble product from two aqueous reactants, and you can identify a precipitation reaction using solubility rules.
Label the components of the double displacement reaction.
CH104: Chapter 5 - Chemical Reactions - Chemistry In this chapter, we will discuss five major categories of chemical reactions: (1) Combination (or Synthesis) Reactions, (2) Decomposition Reactions, (3) Single Replacement Reactions, (4) Double Replacement Reactions, and (5) Redox Reactions. Double Displacement Reaction Definition and Examples - ThoughtCo The reaction between silver nitrate and sodium chloride is a double displacement reaction. The silver trades its nitrite ion for the sodium's chloride ion, causing the sodium to pick up the nitrate anion. AgNO 3 + NaCl → AgCl + NaNO 3 Here's another example: BaCl 2 (aq) + Na 2 SO 4 (aq) → BaSO 4 (s) + 2 NaCl (aq) Answered: Which form of the double displacement… | bartleby Pb (NO3)2 (aq) + 2 KI (aq) → Pbl2 (s) Į + 2 KNO3 (aq) ionic net ionic molecular spectator ions Which form of the double displacement reaction between lead nitrate and potassium iodide is given below? Pb (NO3)2 (aq) + 2 KI (aq) → Pbl2 (s) Į + 2 KNO3 (aq) ionic net ionic molecular spectator ions Question
Label the components of the double displacement reaction.. Displacement reactions | CPD | RSC Education Zinc reacting with lead nitrate: displacement reactions can be some of the most spectacular What students need to know Metals can be placed in order of reactivity in the 'reactivity series'. A more reactive metal will displace a less reactive metal from its compound. Carbon and hydrogen are included in the reactivity series. What are the Parts of a Chemical Equation? | Life Persona A double arrow is used To denote the double meaning of the reaction. Never use an arrow with two ends For this type of reactions since this means that there is resonance, a phenomenon foreign to a chemical reaction (Pastore, 2013). Usually, symbols are often placed on top of the arrow to denote the conditions under which the chemical reaction ... Home Page: The Journal of Arthroplasty Aug 11, 2022 · The Journal of Arthroplasty brings together the clinical and scientific foundations for joint replacement.This peer-reviewed journal publishes original research and manuscripts of the highest quality from all areas relating to joint replacement or the treatment of its complications, including those dealing with clinical series and experience, prosthetic design, biomechanics, biomaterials ... ⚗️What type of reaction is the following chlorine + potassium nitride ... Find an answer to your question What type of reaction is the following chlorine + potassium nitride - nitrogen + potassium chloride ... Double displacement reaction. Balanced equation for the reaction is: KCl(s)+AgNO3(aq)→AgCl(s)+KNO3(aq) ... Label each particle diagram to the type of matter being represented: (element, compound, mixture) a. ...
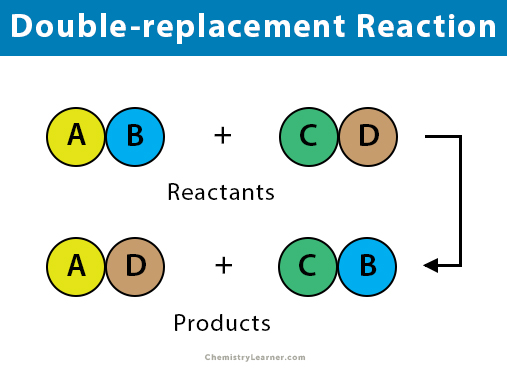
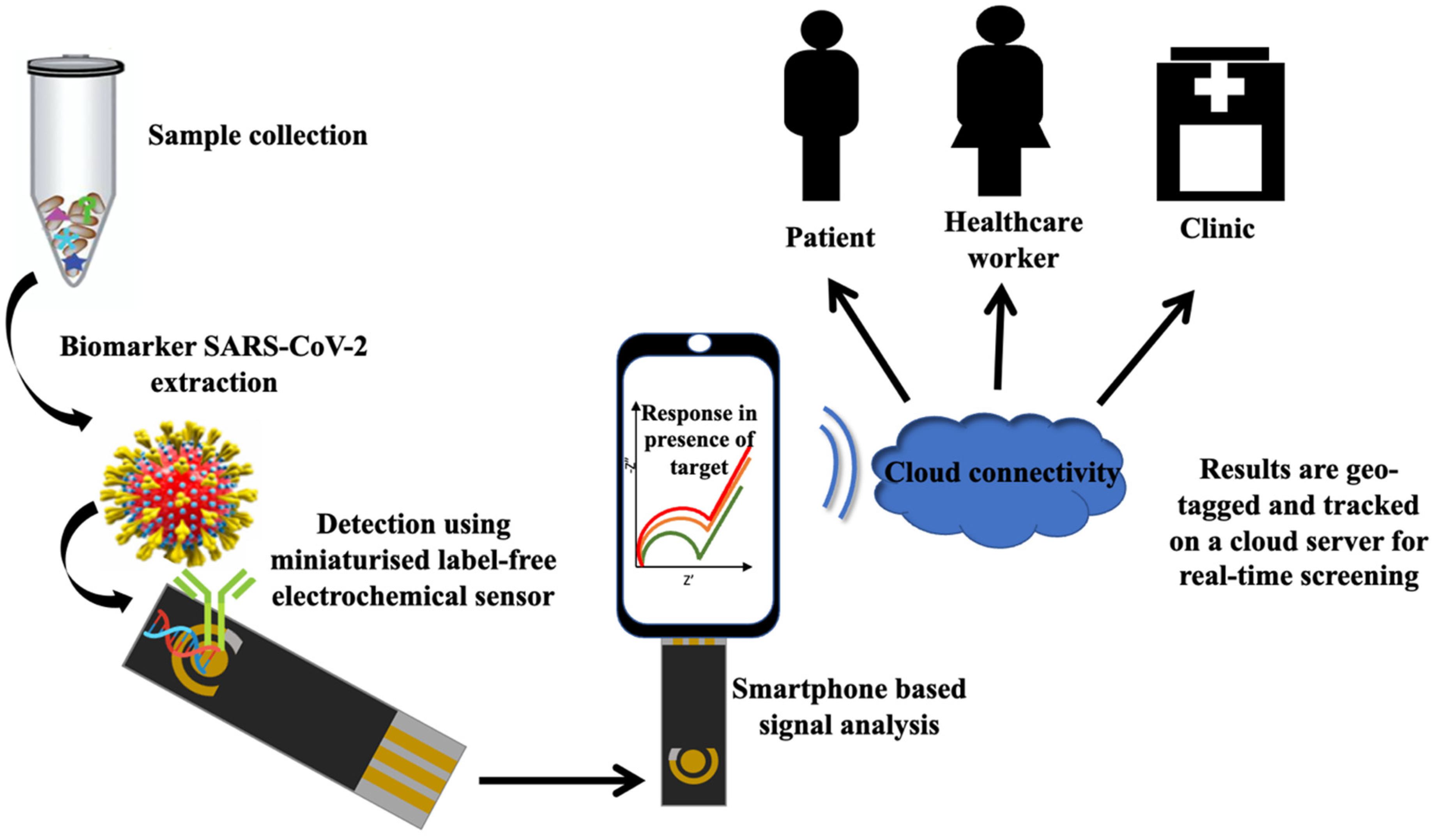
Double Displacement Reaction: Definition and Examples When an element of a compound is displaced by another compound, the reaction is called a single displacement reaction. On the other hand, when elements of two compounds are interchanged between each other, the reaction is a double displacement reaction. These reactions include neutralization, precipitation, and reactions whose products are gases. Chem 11.2 Flashcards - Cram.com Aim: - To Study about types of chemical reactions such as combination, decomposition, displacement and double displacement. - Find out about oxidation and... Essay On Chemical Reactions . Introduction: Chemical reactions are a process that involves, rearranging the molecular structure of substances to create new ones. double displacement reaction Flashcards | Quizlet what is double displacement? exchange of cations in ionic compounds (usually in water) how can you tell if it has occurred? a solid forms (precipitate) a gas is produced water is formed (hard to tell since most of the time reactions occur in water) precipitate a solid that separates from a solution as a result of a chemical reaction Ultrasensitive and label-free electrochemical ... - ScienceDirect Alw I + primer + dNTPs + Exo III + H1 + H2 + MB (note: The concentrations of Exo III, KF polymerase, dNTPs, Nt. Alw I, H1 and H2 were 0.8 U, 250 μM, 10 U, 5.0 μM, 5.0 μM, respectively).
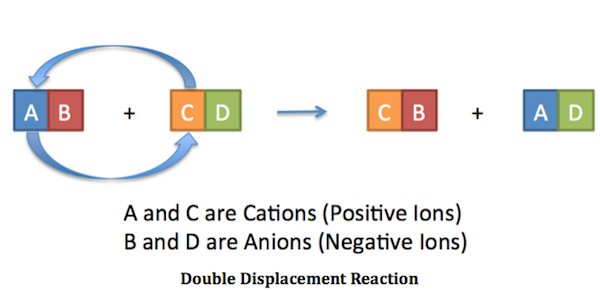
Exp 5 Double Replacement Double replacementreactions (also called "double displacement" or "exchange" or "metathesis" reactions) have the general form AX + BY BX + AY Double replacement reactions typically form a product that is either molecular or ionic. Molecular products such as H 2 O remain in solution and may not appear visually, but gaseous molecular substances 16. Types of Chemical Reactions: Single- and Double ... is an example of a single-replacement reaction. The hydrogen atoms in HCl are replaced by Zn atoms, and in the process a new element—hydrogen—is formed. Solved Question 3 of 3 3. Label the components of the double - Chegg Label the components of the double displacement reaction. BaCl2 (mm) + Na2SO4 (aq) +Ba+2 + Cl + Nat + SO42 - BaSO4 (s) + 2NaCl ( Cations and Anions Ionic Compounds !! Solid and Salt Check Answers ni D Next This problem has been solved! See the answer Show transcribed image text Expert Answer 100% (5 ratings) Hope yo … View the full answer 4.3: Types of Chemical Reactions - Single and Double ... Jun 23, 2022 — A single-replacement reaction replaces one element for another in a ... If a compound is not soluble, we use the (s) label with it and ...
3. Label the components of the double displacement reaction ... Jul 11, 2022 — These are ionic compounds, and here these are ketandanines. This is katine katine and a 9 angetice canines having negative charge and karine ...
7.01: Types of Chemical Reactions - Double Displacement ... May 7, 2020 — 7.01: Types of Chemical Reactions - Double Displacement Reactions · According to the solubility rules, both Ca(NO 3) 2 and KBr are soluble.
CHM131_310_AnionsCationsan... Label the components of the double displacement reaction.1. A cation is an element that _____ one or more electrons to form _____ ions.loses; positiveloses; ...
Answered: Complete and balance the following… | bartleby Answered: Complete and balance the following… | bartleby. Homework help starts here! Science Chemistry Q&A Library Complete and balance the following double displacement reaction: Pb (NO3)2 + NH4OH → Pb2OH Pb (OH)2 Pb (NO3)2 PBOH (NH4)2 (NO3)2 NH4 (NO3)2 NH4NO3 (NH4)2NO3 1 2 4 3.
16.3: Precipitation and the Solubility Product - Chemistry ... Aug 14, 2020 · The ions replace each other based on their charges as either a cation or an anion. This can be thought of as a double displacement reaction where the partners "switching; that is, the two reactants each "lose" their partner and form a bond with a different partner: Figure \(\PageIndex{2}\): A double replacement reaction
Polymerase Chain Reaction Testing: Selected Indications - Aetna The development of the polymerase chain reaction (PCR) has greatly simplified DNA analysis and shortened laboratory time (ACOG, 2002). Polymerase chain reaction allows the exponential amplification of the targeted gene or DNA sequence. Only minute quantities of DNA, typically 0.1 to 1.0 mg, are necessary for PCR.
Molecular convolutional neural networks with DNA regulatory ... Jul 04, 2022 · a, Weight multiplication can be implemented with a simple chemical reaction (left) and its DNA strand displacement process (right).b, The fluorescence kinetics (left) and steady-state fluorescence ...
Neutralization Reaction - Definition, Equation, Examples & Applications A neutralization reaction can be defined as a chemical reaction in which an acid and base quantitatively react together to form a salt and water as products. In a neutralization reaction, there is a combination of H + ions and OH - ions which form water. A neutralisation reaction is generally an acid-base neutralization reaction. Table of Contents
Types of Chemical Reactions: Single ... - BC Open Textbooks is an example of a single-replacement reaction. The hydrogen atoms in HCl are replaced by Zn atoms, and in the process a new element—hydrogen—is formed.
When double displacement reaction of an aqueous solution of ... - Toppr During this reaction, the cations and anions of two different compounds switch places, forming two entirely different compounds. The general equation which represents a double displacement reaction can be written as: Double displacement reactions generally take place in aqueous solutions in which the ions precipitate and there is an exchange of ...
Programmable synthetic cell networks regulated by tuneable ... Jul 06, 2022 · Here we used two different template strands to programme a linear reaction (T 1) and an autocatalytic reaction (T 2) where the product of the linear reaction is required to trigger the ...
To separate the components of a mixture. Science Class-9th Procedure. Step 1: Take the mixture of ammonium chloride, sand and common salt in a china dish. Step 2: Sublimation apparatus is setup as shown in Fig. 1. Step 3: when mixture is heating, Ammonium chloride will separate on the walls of the inverted funnel. Step 4: The residue left behind in the china dish contains sand and common salt.
EXPERIMENT 5 - Double Replacement Reactions - Houston Community College A chemical reaction in which an insoluble product (or precipitate) forms is called a precipitation reaction. The reactants are usually soluble, but the product formed is insoluble and separates out as a solid. Reactions in aqueous solution can be written three ways: 1) as the formula or "molecular" equation 2) as the complete ionic equation
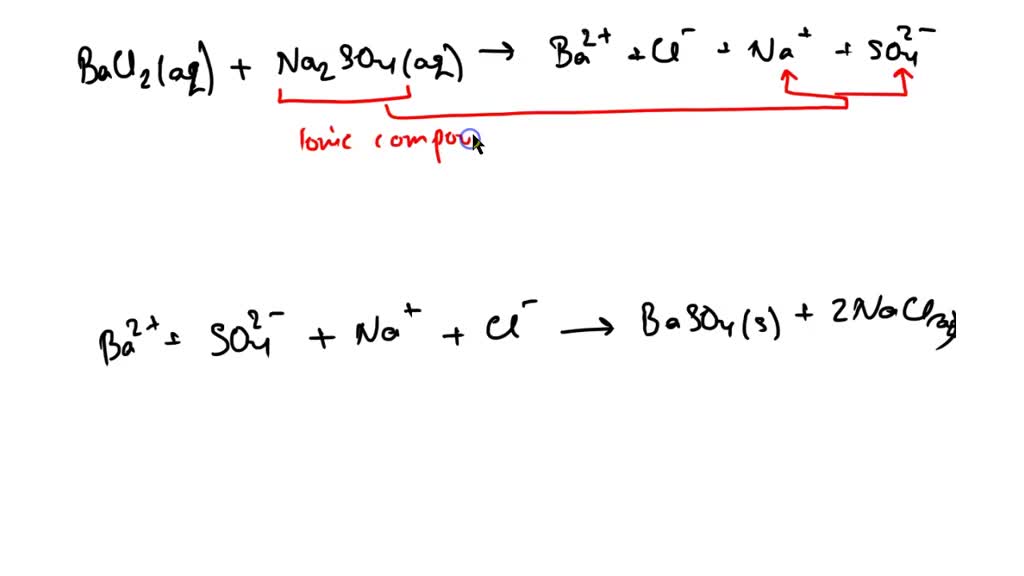
question jof 3 label the components of the double displacement reaction baclztaq nazsohaq ba2 cl nat so basoas 2nacltad calions and anlons ionic compounds solld and salt check answers 03074
CHM101A_AnionsCationsandIonicReactions_162134.pdf - CHM... Label the components of the double displacement reaction. 1. A cation is an element that _____ one or more electrons to form _____ ions. loses; positive loses; negative gains; ... A double displacement reaction occurs when _____. two anions form an ionic bond the reactants, ...
PCR Process Steps Explained - Cole-Parmer May 31, 2022 · Four components (reagents or chemicals) are needed for the PCR process: A DNA or RNA sample (from saliva, blood, hair, skin scraping, etc.) DNA primers: short single-stranded DNA that promotes synthesis of a complementary strand of nucleotides; DNA polymerase: an enzyme that aids in the synthesis of a complementary strand of DNA
How to Identify the 6 Types of Chemical Reactions - Synonym Chemical reactions take place when one set of chemical substances transforms into new ones with different chemical identities. You might observe physical effects, such as light or heat, which might indicate a chemical reaction is occurring. Chemical reactions follow a specific formula, so you can label each reaction ...
Identify the following type of reaction :- •Na2SO4 - Brainly The above chemical reaction is identified as double displacement chemical reaction as two species namely sodium as well as well as barium are displaced. The above chemical reaction is identified as decomposition reaction as the reactant calcium carbonate is splitting into two products namely calcium oxide and carbon dioxide.
Solved Test Your Knowledge Question 3 3 Label the components - Chegg Question: Test Your Knowledge Question 3 3 Label the components of the double displacement reaction Question 3 of 3 Best Naso Ba? + +Na+ + SO BaSO4 + 2Nacio Corpus 11 This problem has been solved! See the answer Show transcribed image text Expert Answer 100% (12 ratings)
2021L2CHEM180DL1_AnionsCationsandIonicReactions_163797.pdf... - Course Hero When an atom loses electrons, it forms a positively-charged particle called a (n). is the term used for any charged particle. Metals tend to form ions with a (n) charge. Anions are particles with a (n) charge.2. Match each term with the best description. Group Metal Cation Ion Positive Negative 3.
chemical reaction and Types of chemical reactions - Blogger Types of chemical reactions, combination reaction, Oxidation and reduction, , Double displacement Reaction, Precipitation reaction, Decomposition rea
Above reaction involves which type of reaction - Toppr Ask The types of reactions are (b) Precipitation and (d) Double displacement. A precipitate of Bariumsulphate is obtained. In double displacement reaction, two compounds react, and the positive ions (cation) and the negative ions (anion) of the two reactants switch places, forming two new compounds or products. So, the correct option is D.
Daylight Theory: SMILES Component parts of a reaction are handled by introducing the ">" character as a new separator. Any reaction must have exactly two > characters in it. ">>" is a valid reaction SMILES for an empty reaction. Each of the ">"-separated components of a reaction must be a valid molecule SMILES. As an aside, molecule SMILES never have a ">" character.
Double Replacement Reaction Definition - ThoughtCo A + B - + C + D - → A + D - + C + B - In this type of reaction, the positive-charged cations and the negative-charged anions of the reactants both trade places (double displacement), to form two new products. Also Known As: Other names for a double displacement reaction are a metathesis reaction or a double replacement reaction .
Anions, Cations, and Ionic Reactions Flashcards | Quizlet True/False: Ionic compounds are chemical compounds in which the anion and cation are held together by strong electrostatic attractions in a lattice structure. True A double displacement reaction occurs when _____. two anions form an ionic bond the reactants, a salt and a solid, share ions the anions and cations of ionic compounds switch places
Answered: Which form of the double displacement… | bartleby Pb (NO3)2 (aq) + 2 KI (aq) → Pbl2 (s) Į + 2 KNO3 (aq) ionic net ionic molecular spectator ions Which form of the double displacement reaction between lead nitrate and potassium iodide is given below? Pb (NO3)2 (aq) + 2 KI (aq) → Pbl2 (s) Į + 2 KNO3 (aq) ionic net ionic molecular spectator ions Question
Double Displacement Reaction Definition and Examples - ThoughtCo The reaction between silver nitrate and sodium chloride is a double displacement reaction. The silver trades its nitrite ion for the sodium's chloride ion, causing the sodium to pick up the nitrate anion. AgNO 3 + NaCl → AgCl + NaNO 3 Here's another example: BaCl 2 (aq) + Na 2 SO 4 (aq) → BaSO 4 (s) + 2 NaCl (aq)
CH104: Chapter 5 - Chemical Reactions - Chemistry In this chapter, we will discuss five major categories of chemical reactions: (1) Combination (or Synthesis) Reactions, (2) Decomposition Reactions, (3) Single Replacement Reactions, (4) Double Replacement Reactions, and (5) Redox Reactions.


























Post a Comment for "42 label the components of the double displacement reaction."